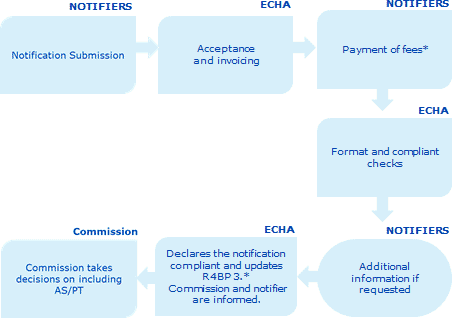
1. General introduction
The aim of this set of Guidance
documents is to gather and to harmonise possible risk
mitigation measures (RMM) for
disinfectants (product type (PT) 1-5). The target group
are all stakeholders working on
authorisations of disinfectants in the biocidal sector (e.g.
applicants, consultants, Competent
Authorities). Several disinfectants are currently
under evaluation within the review
programme established by the Biocidal Products
Regulation (EU) No 528/2012 (BPR)
concerning the placing of biocidal products on the
market. These products represent a
large amount of all biocidal products used in Europe.
To facilitate the work of the
applicants and the Competent Authorities (CA) during the
product authorisation and mutual
recognition, the Guidance documents present a set of
possible RMM that can be used for
all authorisations in Europe and thus simplify mutual
recognitions while ensuring a
similar level of environmental protection.
This Guidance document describes
RMM for drinking water disinfectants to be considered
during the authorisation of
biocidal products as well as the evaluation of active
substances, especially if an
environmental risk is identified. PT 5 disinfectants cover
products used by professional users
as well as by consumers within their outdoor
activities. Drinking water
disinfectants must comply with specific national and European
quality standards set up for water
intended for human consumption. The main types of
disinfection processes are primary
disinfection (main purpose is to kill the majority of
microorganisms), residual
disinfection (maintenance of an anti-microbial potential in the
distribution system), and stand-by
disinfection (high dosage-application to clean up a
contaminated system or when taking
a new system into use). Most of the disinfectants
applied have an oxidizing property
and are not stable. Non-oxidative biocides such as
silver salts or copper/silver
ionisation are used (Emission Scenario Document (ESD) for
PT 5, European Commission 2003).
The Drinking water Directive
98/83/EC requires Member States to ensure that, where
disinfection forms part of the
preparation or distribution of water intended for human
consumption, the efficiency of the
disinfection treatment applied is verified. Thus, also
before the implementation of BPR
most Member States have implemented approval
schemes for drinking water
disinfectants. There are relatively few active substances
applied, among them chlorine,
chlorine dioxide, sodium hypochlorite, and hydrogen
peroxide. Also silver salts,
dichloroisocyanurates, potassium permanganate, and iodine
are applied.
In contaminated distribution
equipment also shock treatments with higher dosages of
disinfectants (e.g. 10 fold of
standard concentration) are required to clean the pipes. In
this case any wastewater generated
should be evaluated and treated, as appropriate,
e.g. by inactivation of chlorine
with sodium thiosulfate
The main emission route of drinking
water disinfectants is to the sewer system and
municipal sewage treatment plants
(STP).
Some of the active substances
and/or other ingredients of the biocidal products are
classified as harmful, toxic or
very toxic to aquatic life and/or may cause long lasting
effects according to Regulation
(EC) No 1272/2008 on classification, labelling and
packaging of substances (CLP
Regulation). Some substances could pose an unacceptable
risk when released to the
environment. If the risk assessment for disinfectant products
results in an unacceptable
environmental risk to aquatic or soil organisms, or to
biological STP (PEC/PNEC > 1)
according to the applicable guidelines these biocidal
products may only be authorised if
the risk can be reduced to an acceptable level by
RMM (conditional authorisation).
In a study on behalf of the German
Federal Environment Agency the existing
environmental RMM for disinfectants
(PT 1-5) proposed by different stakeholders were
resistance of microorganisms through the use of
drinking water disinfectants has
received far less attention. Some publications suggest
that the same mechanisms of
|
|
 Transitional
Guidance on Evaluation of Environmental RMM PT5 Transitional
Guidance on Evaluation of Environmental RMM PT5
compiled and combined to a set of
different RMM that the authorities can choose from
during the product authorisation
process, depending on identified risks. The different
RMM for PT 5 are compiled in the
annex of this document. Considering the progress of
the review programme for existing
active substances, this paper outlines a common
approach for products
authorisations and mutual recognitions.
It should be noted, that there are
RMM which refer to the product designers and
formulators and others which refer
to the user of a biocidal product. The efficiency and
practicability of any RMM to be
quantitatively considered must be evaluated in the risk
assessment by authorities. In this
respect, the possibility of enforcement and control of a
RMM should be considered. Any RMM
referring to the user of a biocidal product must be
clearly indicated on the label.
Only environmental risks from the
use of PT 5 disinfectants are considered in this
guidance document so far.
2. Risk mitigation measures for PT 5 disinfectants
Drinking water disinfectants are an
important tool for maintaining the hygienic quality of
water intended for human
consumption. The use of disinfectants should always be
integrated in a general water
safety plan which includes all steps of water supply from
the protection of the catchment
area to the distribution system. Drinking water
processing consists in
physical-chemical removal processes (e.g. coagulation,
sedimentation, precipitation,
filtration) combined with chemical disinfection, if required.
Drinking water disinfectants
generated in-situ (ozone, chlorine from electrolytic
processes, ultra-violet radiation)
were not covered by the former Biocidal Product
Directive (BPD) but will be
assessed under the new BPR, including possible risks from the
precursor(s).
Most active substances have
oxidation properties and are rapidly eliminated during
application. It is common practice,
to reduce the residual chlorine level to a specific
concentration before the water
enters the distribution system by addition of sulfur
dioxide or sulfite compounds
(dechlorination). During application a part of the oxidative
active substances reacts to
disinfection by-products (DBP) with inorganic or organic
matter present in water. Many DBPs
are harmful and may pose a risk to the environment
and/or form persistent organic
compounds and adsorbable organic halogens (AOX) which
also raise environmental concerns.
Directive 98/83/EC on the quality of water intended
for human consumption, requires
Member States to take all measures necessary to
ensure that any contamination from
disinfection by-products is kept as low as possible
without compromising the
disinfection. The maximum concentration of Trihalomethane
DBPs in drinking water is 100 μg/l (total) and that of Bromate is 10 µg/l, but Member
States are asked to strive for
lower values, where possible without compromising the
disinfection. A background document
on the assessment of DBP is being developed by
CAs where it is inter alia proposed
to carry out PEC/PNEC-assessments of DBP based on
monitoring data from the biocide
uses subjected to authorisation. The results of these
risks assessments should be taken
into account when considering RMM for the respective
products.
There are several proposals for
efficacy testing of drinking water disinfectants . While
the inherent resistance
(susceptibility) of microorganisms and specific pathogens to
drinking water disinfectants has
broadly been analysed, the development of acquired
Transitional Guidance on Evaluation
of Environmental RMM PT5
November 2014
resistance development occur. Resistance development is mainly discussed in
the
context of factors such as
corrosion, dead-end pipes, organic matter, and biofilm
development all supporting the
attachment of microorganisms to surfaces and
preventing their susceptibility to
disinfectants.
Resistance development may be
prevented or reduced by the avoidance of application
faults and of sub lethal
concentrations of the active substances as well as by the use of
alternative substances.
RMM can refer to different
addresses such as the industrial formulator, the supplier and
distributor, the user of
disinfectants, and authorities involved in the surveillance of good
practices.
In this guidance document RMM are
divided in general and specific RMM.
General RMM for example general
precautionary advice, best available techniques, good
housekeeping, applying hygiene
management systems, should be applied to all products,
independent from the results of the
risk assessment, if applicable and exemplify a way to
reduce the use of disinfectants to
the minimum necessary as requested in Article 17(5)
of the BPR. This use shall also
involve the rational application of a combination of
physical, biological, chemical or
other measures as appropriate. They describe
reasonable conditions of use and
reflect common sense. The intention is to avoid
misapplication of disinfectants.
However, general RMM cannot be used in the
environmental exposure assessment
in quantitative terms, because the effect on the
emissions and the compliance cannot
be proven.
Specific RMM result from the risk
assessment and are suitable for a quantitative
reduction of the exposure through
modification of the respective emission scenarios.
Note that RMM for users have to be
clearly communicated with the label or product
leaflets. Specific RMM are designed
to reduce an identified environmental risk (PEC/PNEC
> 1) to an acceptable level. The
efficiency and practicability of specific RMM has to be
proven by the applicant for
authorisation of a biocidal product by submitting sound data
or studies. Some RMM might also be
appropriate if the risk quotient shows a level of
concern (e.g. PEC/PNEC > 0.1).
This may for example, be the case if a substance is used
in different PT simultaneously.
Specific RMM should be considered in the revision of
Emission Scenario Documents (ESDs)
as far as possible in order to harmonise the
approach. If they represent the way
the product is commonly applied, the efficiency of
the RMM could be quantified.
4.1 Categorisation of specific RMM
Specific RMM can be attributed to
different categories described below. The precise RMM
for each category and specific
unacceptable risks can be found in the Appendix I of this
document. It should be noted that
some RMM, whose main focus is on human health,
nonetheless indirectly lead to
lower exposure to the environment, e.g. because specific
uses or user categories are
excluded. These are also included in the document.
e.g. Shrivastava R et al. 2004. Suboptimal chlorine treatment of drinking water leads to selection of
multidrug-resistant Pseudomonas aeruginosa. Ecotoxicol
Environ Saf. 58(2):277-283
Transitional Guidance on Evaluation
of Environmental RMM PT5
Drinking water disinfectants are
mainly applied by specifically trained professional users
such as drinking water operators.
On a smaller scale privately owned treatment plants
for outlying settlements exist next
to mobile drinking water tanks from outdoor
activities.
The benefits of consumer use of PT
5 disinfectants should carefully be compared with the
feasibility of non-chemical
treatment techniques. With respect to RMM for consumer uses
of disinfectants only short and
simple instructions are likely to be implemented by the
user. Thus, emphasis should be on
product integrated RMM under the control of the
supplier (chemical composition and
design). The product label should communicate all
instructions on safe use, storage
and disposal to consumers. These instructions are
mainly attributed to general RMM
which cannot be quantitatively assessed.
To exclude non-professional
(consumer) uses of PT 5 disinfectants, a measure could be
taken for these disinfectants not
to be offered on open shelves or by internet commerce
through self-service.
Drinking water disinfectants are
mainly applied in public or industrial drinking water
abstraction plants but there exist
also (very) small water supplies from private owners.
Additionally mobile disinfection
devices exist for the outdoor sector. The water source
often determines the quality of the
water. Groundwater sources generally are of superior
quality to surface water from
rivers and reservoirs and require less treatment. The area
of use and the choice of the water
source may also contribute to reduce the formation of
DBP through the use of some
oxidative disinfectants, e. g. by avoiding areas where the
inorganic or organic precursors of
such DBP are known and present.
The practicability of RMM
concerning the area of use depends on the unambiguous
description of allowed uses.
Because the intended uses determine the emission scenarios
to be assessed, these RMM may be
considered in quantitative terms.
In most cases the biocidal product
is identical to the active substance or its precursor.
The possible formation of DBPs
should also be considered.
PT 5 disinfectants are mainly
applied by automatic dosing pumps. In certain
circumstances the disinfectant is
manually added to a water tank, especially within
outdoor activities. Accurate dosage
is one factor to prevent risk for the environment and
avoid spillages. The possible
formation of DBPs should also be considered when
evaluating the formulation. Product
integrated RMM may be quantitatively considered in
the exposure assessment.
4.1.5 Packaging and pack size
The packaging of the product also
plays a role and can be used to reduce environmental
exposure by avoidance of over
dosage and disposal of unused product. Product designs
supporting the application of
disinfectants through accurate dosing, e.g. via dosing
pumps should be preferred.
Therefore, where appropriate, the placing on the market
should be restricted to certain
specific product design.
Product integrated RMM may be
optimized by product developers and discussed with
authorities. They could be
considered in the exposure assessment in quantitative terms if
appropriate. It is recommended to
develop an overview of CE marked labelled devices.
At present it is not clear in what
extent specific devices would lower the use and thus
  Transitional
Guidance on Evaluation of Environmental RMM PT5 Transitional
Guidance on Evaluation of Environmental RMM PT5
November 2014
emission of the biocidal product to
a safe level for the environment. It would be helpful if
more information would become
available for environmental risk assessment.
4.1.6 Treatment and/or disposal
The main emission pathway for PT 5
disinfectants is via the sewer system. The removal
of precursor of DBP and
disinfection concentrations exceeding the limit values by
technical treatment and the removal
of DBP before the water enters to the distribution
system are possible options for
RMM. These RMM may only be considered in quantitative
terms in the exposure assessment if
they are implemented in routine practice by the
user and if some surveillance is
carried out by authorities.
Article 69 (1) of the Biocidal
Products Regulation (EU) No 528/2012 stipulates that
biocidal products shall be labelled
in accordance with the SPC, and with Directive
1999/45/EC relating to the
classification, packaging and labelling of dangerous
preparations, and where applicable
Regulation (EC) No 1272/2008. This includes
precautionary statements. However
the requirements of these legislations may not allow
a sufficient description of
possible specific risks which may arise during the use of
disinfectants and be detected
during the risk assessment. Therefore, additionally
standard phrases should allow a
sufficient description of the special risks and of the
safety precautions to be taken where risks have been identified. Thus, in
addition to the
elements already listed in Article
69(2), product labels or the packaging of disinfectants
should show the safety precautions
for the protection of humans, animals or the
environment. These safety
precautions should always be carried on the label of the
products or on an accompanying
leaflet together with the other directions for use and
disposal of the product. Reference
only to an internet source is not sufficient.
4.1.8 Codes of Good Practices
The careful use of disinfectants is
essential to minimise risks for human health and the
environment. In many application
areas for disinfectants good and best practice
documents and training courses have
been developed. Maintaining good water
processing practices is a
prerequisite for disinfectants being effective. The design of the
equipment and the facility helps
minimising the amount of disinfectant. Several good and
best practice documents as well as
technical standards cover the processing of drinking
water and minimisation of the
formation of DBPs. Some non-exclusive examples are:
• White, G. C. 2010. White’s handbook of chlorination and alternative
disinfectants.
5th Edition,
Black & Veatch Corporation, John Wiley & Sons, Hoboken, New
Jersey.
• Niessner,
R., Höll, K. 2010. Wasser Nutzung im Kreislauf: Hygiene, Analyse und
Bewertung. 9th edition, De
Gruyter, Berlin.
• Dammers, N. 2011. Towards a Guidance Document for the implementation of
a
Risk Assessment
for small water supplies in the European Union - Overview of
best practices.
Study of the Water cycle Research Institute on behalf of DG ENV
European
Commission, November 2011.
• WHO 2011. Guidelines for Drinking-water Quality - 4th Edition. World
Health
Organisation,
WA 675, Geneva, Switzerland.
• Le Chevallier, M. W., Au, K. K. 2004. Water Treatment and Pathogen Control:
Process
Efficiency in Achieving Safe Drinking Water. World Health Organization,
This is by analogy to what has been done in the PPP area where standard
phrases for special risks and safety
precautions for
plant-protection products have been established.
Transitional Guidance on Evaluation
of Environmental RMM PT5
• WHO, OECD. 2003. Assessing microbial safety of drinking water -
Improving
approaches and
methods. IWA-publishing, London.
• Weinberg, H. S., Krasner, S. W., Richardson, S. D., Thruston, A. D.,
2002. The
Occurrence of
Disinfection By-Products (DBPs) of Health Concern in Drinking
• US EPA 1999. Alternative Disinfectants and Oxidants Guidance Manual.
United
States EPA
Environmental Protection Agency. EPA 815-R-99-014, April 1999.
• Borchers, U. 2012. Die Trinkwasserverordnung 2011: Erläuterungen- Änderungen
– Rechtstexte. Beuth-Verlag Berlin.
The drinking water guideline of the
WHO and supporting documents as well as the
drinking water guideline of the
European Commission are supplemented by national
guidelines and lists of approved
drinking water disinfectants. The
formation of DBP
could partly be managed by
avoidance and/or removal of the inorganic or organic
precursors.
In addition to product labelling
and instructions for use, several good and best practice
documents should be made available
to the user.
RMM referring to codes of good
practice may only be considered in quantitative terms in
the exposure assessment if these
good practices are well established in professional use
of disinfectants and if some
surveillance by authorities is carried out. The practicability of
these RMM is not under the control
of the authorisation process for disinfectants. RMM
regarding good practices do not
apply for consumer use of disinfectants.
Transitional Guidance on Evaluation
of Environmental RMM PT5
November 2014
In this annex RMM for products used
in the PT 5 are proposed.
The named general RMM should be
applied to all products, if suitable, to ensure a proper
and safe use of biocidal products
throughout the life cycle when their use is needed.
Words written in italic font in brackets should be
adapted respectively for each
application of the biocidal
product. They are only placeholders and illustrate proposals.
Depending on the application of the
disinfectant the sentences can be chosen and/or
modified. The Precautionary
Statements of the CLP Directive and the label requirements
according to Article 69(2) of the
BPR are not repeated here but have to be followed.
• Take care for general good hygiene and good water processing practice.
• Examine whether the use of disinfectants can totally or partially be
substituted by
other (e.g.
microfiltration) processes.
The following specific RMM can be
chosen based on identified unacceptable risks during
the risk assessment. The RMM are
assigned to tables related to the first environmental
compartment whereto the substance
is released. In most of the cases for disinfectants
this is the STP. These RMM can also
have an effect on possible unacceptable risks in the
following compartments (e.g. a
measure that lowers the concentration in the influent of
the STP can also lower the
concentration in the receiving surface water after the STP).
RMM suitable for other cases where
the substance is directly released to other
compartments are arranged in tables
as well as relating to the receiving compartments
below. Some specific RMM might be
too difficult to be followed by non-professional
users. Thus, emphasis for these
products should be on product integrated RMM under
the control of the supplier
(chemical composition and design, packaging, etc.).
Words written in italic font in brackets should be adapted
respectively for each
application of the biocidal
product. They are only placeholders and illustrate proposals.
The list is not exhaustive and
should be continued during the product authorization
process.
Example 1: Risk in the STP
If during the risk assessment for a
disinfectant a risk is identified for the STP the risk
assessor can use a RMM from Table 1
(Possible RMM for unacceptable risks associated
with the direct release to the
STP). These RMM describe possible ways to mitigate risks.
Not all RMM are suitable for each
case, the decision on what RMM to choose and how to
modify it has to be made
case-by-case.
Example 2: Risk in surface water
A risk in surface water can result
from a direct or an indirect exposure. If the risk is due
to an indirect exposure through the
STP the risk assessor could use a RMM from Table 1
(Possible RMM for unacceptable
risks associated with the direct release to the STP) to
mitigate the risk. If the risk is due to a direct exposure the risk
assessor could use a
RMM from Table 2 (Possible RMM for unacceptable risks associated with the direct
release to surface water). Again, the choice of the RMM has to be based on the
application of the product and
should be feasible.
ECHA.EUROPA.EU
|